What Would You Change?
The Diagnostic Breakthrough
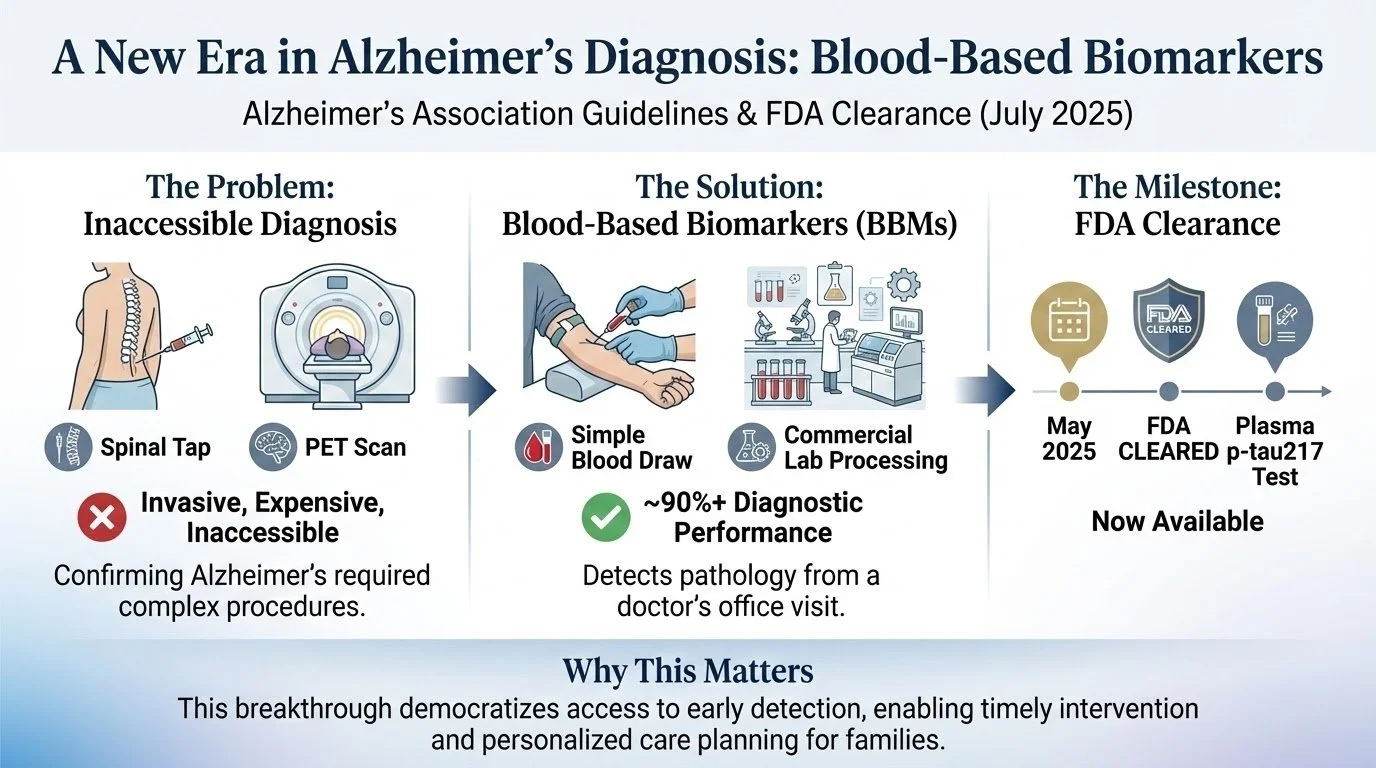
On July 28, 2025, the Alzheimer's Association released clinical practice guidelines recommending how specialty clinicians should use blood-based biomarkers (BBMs).
Why this matters: Until now, confirming Alzheimer's disease required either a lumbar puncture (spinal tap) to analyze cerebrospinal fluid or a PET scan. Both are invasive, expensive, and inaccessible to many families. Now, a simple blood test—drawn at your doctor's office, processed by commercial labs—can detect Alzheimer’s pathology with ~90%+ diagnostic performance in peer-reviewed studies (exact performance varies by assay and population). In May 2025, FDA cleared the first blood test measuring plasma p-tau217 and it is now available.
Do you want to know?
The Question No One Is Asking
Doctors are asking:
"Should we order the test?"
Researchers are asking:
"How accurate is it?"
Insurance companies are asking:
"Will we cover it?"
But the most important question for families is:
What would you change if you knew you had Alzheimer's pathology in your brain?
Not "what can be done"—what would YOU do differently?
What It Means to Know
You have Alzheimer's pathology—amyloid plaques and tau tangles—accumulating in your brain right now.
It does NOT mean:
You will definitely develop dementia (some people with pathology never progress)
You will decline rapidly (timeline is unpredictable—could be 2 years, could be 20)
Your fate is sealed (interventions can slow progression)
You should panic
What it DOES mean is that a door has opened to early intervention when new treatments are available and when lifestyle intervention works best. A door to legal planning while you have full capacity. A door to decisions that become more difficult later. A door to preparing your family before crisis.
The New Disease-Modifying Therapies
Lecanemab and donanemab—FDA-approved drugs that slow Alzheimer's progression by about 25%—have strict eligibility requirements:
✅ Mild cognitive impairment OR mild dementia
✅ Confirmed amyloid pathology (requires biomarker test)
❌ NOT moderate or severe dementia
Translation: If you wait until symptoms are obvious, you're likely ineligible. The treatment window closes at moderate dementia.
The Lifestyle Intervention Evidence
The FINGER trial in Finland and US POINTER trial (2025) proved that intensive multi-domain lifestyle interventions can slow cognitive decline. The Lancet Commission (2024) reported 14 modifiable risk factors for dementia. Higher physical activity is associated with slower cognitive and functional decline in individuals with elevated baseline amyloid. Critical finding: Benefits are greatest when started in the MCI stage or earlier. Once moderate dementia arrives, lifestyle interventions are much less effective for patients, but become more important for managing caregiver stress and well-being.
Approaches Providers Can Take Right Now
Approach 1: "Test Everyone with Symptoms"
The problem: Testing without infrastructure to support results. Knowledge without guidance. Diagnosis without stewardship.
Approach 2: "Don't Test Unless It Changes Management"
The problem: Avoidance doesn't prevent progression—it just prevents preparation. The "wait and see" approach guarantees missing treatment windows.
There's a Third Approach: Decision-Preparedness Testing
The question isn't "should we test?" The question is: "What would we do with results?"
"If the test shows Alzheimer's pathology, what would you do?"
Would you pursue disease-modifying therapy?
Would you intensify lifestyle interventions?
Would you accelerate legal and financial planning?
Would you have family conversations about the future?
Would you consider clinical trial participation?
"What are your biggest fears about knowing?"
Anxiety about the future?
Impact on relationships or self-identity?
Discrimination concerns (employment, insurance)?
Not knowing how to act on results?
If your list has 3+ items, have a conversation about testing. Tests are best acted on in specialty care or within a defined care pathway, per the AA guideline.
What To Do Next
If you or a family member has cognitive concerns:
1. Seek Expert Evaluation (Not Just Primary Care Screening)
Primary care doctors are excellent at many things, but detailed cognitive assessment and BBMs interpretation require specialized expertise
See a neuropsychologist, neurologist, or geriatrician with cognitive disorders expertise
Insist on comprehensive evaluation, not just MoCA or MMSE (too insensitive for early changes)
2. Don't Go It Alone
Cognitive decline is a family journey—involve spouse, adult children, trusted friends
Seek services that provide longitudinal support, not just episodic evaluation
Consider programs like Cognitive Stewardship that offer continuous monitoring and decision coaching
3. Act on Results
If biomarker-positive: Immediately begin evidence-based interventions (lifestyle optimization, DMT evaluation if appropriate, advance planning)
If biomarker-negative: Pursue alternative diagnoses and address modifiable factors
Don't let positive results cause paralysis—this is where expert guidance is critical
4. Stay Informed
Biomarker science is evolving rapidly—guidelines will update
Clinical trials are ongoing—opportunities for cutting-edge treatment
New therapies in pipeline—tau-targeting drugs, anti-inflammatory agents, combination therapies
The window for intervention is open. The tools for early detection are here. The question is: Will you use them?
Early Detection Is Only Valuable If Followed by Action
The 2025 Alzheimer's Association guidelines on BBMs represent a pivotal moment in the fight against Alzheimer's disease. For the first time, we can detect pathology early, accurately, and accessibly.
Some experts believe direct-to-consumer or at-home Alzheimer’s blood biomarker testing may be “an innovation too far” — meaning it may cause more harm (anxiety, misinterpretation, unnecessary follow-up tests, discrimination) than benefit in the current state.
They want:
Better care infrastructure: Legal protections against discrimination for biomarker-positive individuals; more clinicians trained in dementia/Alzheimer’s care.
Funding & workforce expansion: More specialists need to be trained and available.
Verified treatments in asymptomatic people: Tests only make sense when there are actionable interventions for people who test positive but do not have symptoms.
This is why I created Cognitive Stewardship. The science of early detection has advanced faster than the clinical infrastructure to support it. Families are getting biomarker results without guidance to know what’s next.
We close that gap.
If you or a loved one has cognitive concerns, or if you've received biomarker results and aren't sure what to do next, Treasure Coast Cognition is here to help.
References
Alzheimer's Association. Appropriate Use of Blood-Based Biomarkers in Alzheimer's Disease: Clinical Practice Guideline From the Alzheimer's Association. Alzheimers Dement. 2025. Published online July 28, 2025. doi:10.1002/alz.14225
Labcorp. Labcorp Launches First FDA-Cleared Blood Test for Alzheimer's Disease. Published May 2025.
Yaffe K, Barnes DE, Rosenberg D, et al. Effect of a Multidomain Lifestyle Intervention vs Health Education on Cognitive Function in Older Adults at Increased Risk of Alzheimer Disease: The US POINTER Randomized Clinical Trial. JAMA. 2025;333(3):217-229. doi:10.1001/jama.2024.24157
Livingston G, Huntley J, Liu KY, et al. Dementia Prevention, Intervention, and Care: 2024 Report of the Lancet Standing Commission. Lancet. 2024;404(10452):572-628. doi:10.1016/S0140-6736(24)01296-0
Rabin JS, et al. Physical Activity and Longitudinal Cognitive and Functional Decline in Alzheimer Disease. Nat Med. 2025. doi:10.1038/s41591-025-03955-6
Personalized Prevention: Targeting Modifiable Risk Factors in Cognitive Stewardship
2024 Lancet Commission: 45% of Dementia is Potentially Preventable in the Population
The 2024 Lancet Commission on dementia prevention now identifies 14 modifiable risk factors that collectively account for 45% of dementia cases worldwide—up from 40% in the prior analysis with the addition of high LDL cholesterol and vision loss.
45% represents the theoretical maximum if all risk factors were eliminated across entire populations from early life through late life. This does NOT mean that any individual optimizing their risk factors has a 45% reduced dementia risk.
Midlife Interventions May Be Most Critical
The majority of modifiable risk burden (approximately 60% of the 45%) occurs in midlife when vascular and metabolic risk factors exert their greatest impact on brain health decades later.
Life-Course Risk Factor Model
Early Life (ages <45):
Less education (5%) - Limited cognitive stimulation and cognitive reserve building in formative years
Midlife (ages 45-65):
Hearing loss (7%) - The single largest modifiable risk factor
High LDL cholesterol (7%) - NEW for 2024; midlife dyslipidemia accelerates vascular brain changes
Depression (3%) - May be both risk factor and early symptom
Traumatic brain injury (3%) - Even mild TBI increases long-term dementia risk
Physical inactivity (2%) - Sedentary lifestyle deprives brain of neurotrophic benefits
Diabetes (2%) - Chronic hyperglycemia damages cerebral microvasculature
Smoking (2%) - Vascular damage and oxidative stress
Hypertension (2%) - Uncontrolled BP in midlife drives white matter disease
Obesity (1%) - Metabolic syndrome and chronic inflammation
Excessive alcohol (1%) - >21 units/week associated with brain atrophy
Late Life (ages >65):
Social isolation (5%) - Lack of cognitive stimulation and increased depression risk
Air pollution (3%) - Fine particulate matter (PM2.5) exposure causes neuroinflammation
Vision loss (2%) - NEW for 2024; untreated cataracts and poor vision reduce engagement
2025 US POINTER Trial: Lifestyle Prevention
Published January 2025 in JAMA, the US Study to Protect Brain Health Through Lifestyle Intervention to Reduce Risk (POINTER) trial represents the first large-scale validation of the Finnish FINGER model in Americans.
Key Findings
Primary Outcome - Global Cognitive Composite: equivalent to 1-2 years of preserved cognitive function
Critical Insights: Any lifestyle intervention, even minimal, provides benefit. However, structured programs deliver ~30% greater cognitive gains (0.24 vs. 0.21 SD/year improvement). The program works, but only if people actually do it. Cognitive Stewardship provides care coordination and evidence-based strategies adherence strategies for maintenance of behavior change to slow decline, not prevent dementia.
Cognitive Stewardship can help translate the Lancet Commission's 14 modifiable risk factors and the US POINTER trial's structured intervention model into a personalized, sustainable program for your brain health. Through continuous monitoring, expert guidance, and intensive support, we can help you implement these evidence-based strategies proven to slow cognitive decline—while also navigating the 10 critical decision points where expert guidance prevents costly mistakes. You get both: (1) Personalized lifestyle intervention for cognitive protection, and (2) Decision-preparedness coaching for life transitions. Together, these maximize your cognitive trajectory and quality of life.
References
Livingston G, Huntley J, Liu KY, et al. Dementia Prevention, Intervention, and Care: 2024 Report of the Lancet Standing Commission. Lancet. 2024;404(10452):572-628. doi:10.1016/S0140-6736(24)01296-0
Yaffe K, Barnes DE, Rosenberg D, et al. Effect of a Multidomain Lifestyle Intervention vs Health Education on Cognitive Function in Older Adults at Increased Risk of Alzheimer Disease: The US POINTER Randomized Clinical Trial. JAMA. 2025;333(3):217-229. doi:10.1001/jama.2024.24157
The Healthcare System Isn’t Ready
A colleague was at her primary care visit. She asked how to get blood-based biomarker testing at a major university medical center (where she sees her doctors), and was told that she could try to go through neurology but at her age it was basically impossible. Maybe the response would have been different if she were older, but it struck her that even those proactive about getting tested face significant barriers.
Even at top medical centers, proactive families face impossible barriers:
You can't get blood biomarker testing because you're "too young" or "not symptomatic"—despite tests that predict dementia 10 years before symptoms appear.
Your primary care doctor waits for obvious memory problems before acting—by then, treatment eligibility windows have already narrowed dramatically.
Specialist referrals take 3-6 months. Eligibility testing takes another 2 months. By the time traditional care pathways respond, half of early dementia patients have already progressed beyond treatment eligibility.
Healthcare system delays happen when you need action.
Cognitive Stewardship changes this.
We help families act years earlier—with proactive monitoring, personalized intervention, and access when concerns emerge—so you can intervene when science shows it matters most.
The Waiting Years
The Waiting Years.
A friend reached out.
She told me about her mother.
October 2023: The family noticed changes. Memory issues. Movement struggles. They called the university memory clinic — the one everyone recommends.
First available appointment: September 2024.
So they found a physician’s assistant in another city. Tried there. The physician’s assistant was doing their best. The answers didn’t fit what the family was seeing.
By the time they finally reached the specialist and got a DaTscan ordered, it was August 2025.
How hard is it to get imaging?
MRI: Easy.
DaTscan: Not easy.
Real-world barriers:
• Insurance approval: weeks to months
• Limited centers
• 6–12 month wait times
• Some neurologists wait until symptoms are “obvious enough”
Meanwhile, families wait.
Nearly two years from first noticing changes to getting the right diagnosis.
Not because they didn’t care.
Not because they didn’t try.
Because the system isn’t built for the early stage.
In those two years, they navigated alone:
Is this normal aging?
Should we push for different tests?
Which supplements matter?
When do we get a walker? A shower chair?
How do we find the right support?
Where do we find support?
My friend is a researcher. She did everything right. She dug through forums, found Zoom groups, paid thousands for a “personalized” program that delivered generic guidance.
She told me:
“I would have paid anything to not wait. Not for a cure — just for someone who knew what to ask for and when.”
That gap — between noticing something is wrong and getting meaningful support — is where families lose time.
Cognitive Stewardship™ can help.
Not to replace your medical team.
Not to diagnose alone.
To guide families through the waiting — the uncertainty, the decisions, the system navigation.
To make sure that when they finally reach the specialist, they haven’t lost two years.
If you’ve lived this — the waiting, the confusion, the “I wish someone had told us” — what did you need most?
I’m building this for the families who shouldn’t have to navigate alone.
Please share your story. Your experiences are shaping this work.
The views expressed here are my own and do not represent those of the NIH or the U.S. government.